Introduction Of UV Absorbers For Cosmetics
Leave a message
● In 1928, Phenyl Salicylate and Phenyl Cinnamate are used for emulsion cosmetics in the
United States.
● In 1930, Benzyl Salicylate additive appeared in Australia.
● In 1996, Avobenzone was approved as a Class 1 breed.
● In 1998, Zinc Oxide was approved.
● In 1998, Ecamsule was approved.
Main UV absorber list
Number | Types | Structure | Commercial Products | wavelength |
1 | Para-aminobenzoic acid |
| Octyl dimethylaminobenzoate、Para-aminobenzoic acid | 280-330 nm UVB |
2 | Salicylate |
| Octyl Salicylate、Homosalate、Phenyl salicylate and Benzyl salicylate etc. | 280-320 nm UVB |
3 | Cinnamate |
| Octyl Methoxycinnamate、IsoaMyl 4-MethoxycinnaMate etc. | 280-330 nm UVB |
4 | Benzophenone Series |
| Benzophenone(BP)、 Benzophenone-1/2/3/4/6/9 | 280-330 nm UVB |
5 | Camphor Derivative |
| 3-(4'-Methylbenzylidene) Camphor、Ecamsule | 330-360 nm UVA |
6 | Dibenzoylmethane |
| Avobenzone | 356 nm |
7 | Benzotriazole |
| Bisoctrizole、UV-326、UV-327、UV-328、UV-329 and UV-P etc. | 280-380 nm |
8 | Triazone |
| Bemotrizinol、T-150 and UV-1577etc. | 280-380 nm UVA+UVB |
9 | Acrylonitrile |
| Etocrylene etc. | 280-330 nm |
1.1 Para-aminobenzoic acid
P-aminobenzoic acid obtained a patent very early and is a class of UV absorbers based on p-aminobenzoic acid. It is crystallized by hydrogen bonds between molecules, is easily oxidized in the air, and has high water solubility. The effectiveness and stability of the analysis can be changed by a certain modification of the functional group and the para-aminobenzoic ultraviolet absorber can be applied more widely. The two kinds of para-aminobenzoic acid ultraviolet absorbers widely used are p-aminobenzoic acid and 4-ethylhexyl 4-dimethylaminobenzoate in China. The structure is shown in Figure 1-1 and Figure 1-2:

1.2 Salicylate
Salicylate is the first UV absorbers to be used in sunscreens. Over the years, salicylate UV absorbers have been produced in large quantities and their widespread use has benefited from its milder and more stable properties. At the same time, it has better safety than other UV absorbers. Of course, the disadvantage is that its absorption rate of ultraviolet light is relatively low. The salicylate UV absorber comprises phenyl salicylate, amyl salicylate, p-isopropyl isopropyl salicylate, 4-isopropyl benzyl salicylate salicylate and humulizate. Wait. Figure 1-3 shows the structural formula of salicylate UV absorbers:

Figure 1-4 4-Methoxycinnamic acid-2-ethylhexyl ester structure
1.4 Benzophenone Series
Benzophenones are important UV absorber with a maximum wavelength of around 330 nm. They are moderately absorbing UVB and UVA bands and have been widely used. Since benzophenone-based UV absorbers have been found to have hidden cancer-causing hazards in recent years, there are not many types of sunscreen-based cosmetics, and their structural formulas are shown in Figure 1-5:

Figure 1-5 Structural formula of benzophenone UV Absorbers
1.5 Camphor Derivative
Camphor derivative UV absorbers have perfect UV absorption efficiency. Over the years, through the molecular design of camphor derivatives, a camphor derivative UV absorber with strong absorption rate has been synthesized. It is one of the most widely used UV absorbers in China. Commonly used such as 3-(4'-Methylbenzylidene)-d-1-camphor, 3,3'-(1,4-phenylenediethylene)Bis (7,7-dimethyl-2-oxo-2-cyclo[2.2.1] Hept-1-yl methanesulfonic acid), the structure is shown in Figures 1-6 and Figure 1-7:

Figure 1-6 3-(4'-Methylbenzylidene)-d-1-camphor
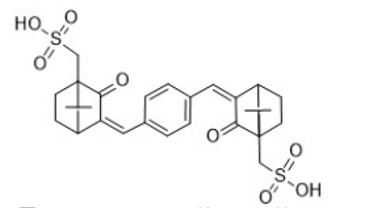
Figure 1-7 3,3'-(1,4-phenylenediethylene)Bis (7,7-dimethyl-2-oxo-2-cyclo[2.2.1] Hept-1-yl methanesulfonic acid)
1.6 Dibenzoylmethane
Dibenzoylmethane-based UV absorbers are a kind of fat-soluble UVA absorbers due to the strong absorption of ultraviolet rays in the range of 320-400 nm in tautomerization due to the presence of keto-enol tautomers. Among them, p-tert-butyldibenzoylmethane is an UV absorber developed by Swiss Givaudan Company in the 1980s. It is widely used in daily chemicals, plastics, coatings, polymers, etc. It is currently the only long-wave UV absorber approved by the US FDA. Its enol structure has large absorption at 345 nm and its keto isomer has large absorption at 260 nm. It is a perfect UVA ultraviolet absorber, but its photochemical stability is low, including Butyl methoxydibenzoylmethane, cresoltrizole trisiloxane, polysiloxane-15 etc. Avobenzone can absorb most of the wavelength range of UVA and it is also the most effective one of the few UVA absorbers in the world. Its structural formula is shown in Figure 1-8:

Figure 1-8 Avobenzone structure
1.7 Triazone
Triazines are a class of macromolecular new UV absorbers developed in recent years. These UV absorbers have high UV absorption efficiency. Some of these UV absorbers have wide absorption band and absorb both UVA and UVB. In the band of ultraviolet light, their ultraviolet absorption mechanism is intramolecular proton transfer (ESITP). At the same time, since the molecular structure of the triazine-based ultraviolet absorber is too large, solubility is also a problem to be solved. Its structural formula is shown in Figure 1-9:

Figure 1-9 Structural formula of triazine UV Absorbers
1.8 Benzotriazole
Phenylbenzimidazole UV absorbers are a class of UV absorbers that have developed rapidly in recent years.2-Phenylbenzotriazole-5-sulfonic acid has perfect water solubility and high ultraviolet absorption efficiency is widely used in water-soluble cosmetics.Such UV absorbers have high molar absorption coefficient in the range of 300-385nm and the ultraviolet absorption mechanism is also intramolecular proton transfer (ESITP). Its structural formula is shown in Figure 1-10:

Figure 1-10 Structural formula of benzotriazole UV Absorbers
1.9 Structural compound class
Representative of structural composite UV absorbers - Diethylamino Hydroxybenzoyl Hexyl Benzoate, referred to as DHHB, is the latest UVA sunscreen ingredient and is an oil-soluble chemical sunscreen that absorbs ultraviolet light with a wavelength of 320-400 nm. Its maximum absorption peak is at 354 nm and its absorption performance for UVA is almost the same as that of avobenzone, but DHHB has very high stability compared with avobenzone. Its structure is shown in Figure 1-11:

Figure 1-11 Diethylamino Hydroxybenzoyl Hexyl Benzoate structure
Currently, among the chemical sunscreen products in the UVB band, isooctyl salicylate, humulizate, isooctyl p-methoxycinnamate, octocrylene, benzophenone-3,benzophenone-4
are basically the mainstream compound choice.They are used in large quantities, but the disadvantage is that the absorption is not good enough.

Ethylhexyl triazinone is a prominent representative of the triazine series, with good absorption and good applicability.

The basic requirements for all UV absorbers used in sunscreen are: 1) efficacy; 2) safety; 3) registration; 4) free operation of intellectual property status. The development of effective molecules is prerequisite before other aspects work. First, efficacy means good absorbance in the most interesting spectral range of sunscreens from 290 to 400 nm. It also means that a sufficient amount of material can be incorporated into the cosmetic formulation. The UV absorber can be dissolved in the oil phase or the aqueous phase of the sunscreen formulation, so the respective solubility must be sufficiently high. Another possibility is to use a dispersion of fine particles of absorbing material. In all countries, new UV absorbers require pre-market approval. When testing animals with extensive toxicology testing procedures, including in the human body, it must be demonstrated that the new UV absorber is safe. So far, the European scientific security perspective published by the Consumer Safety Science Council has become the benchmark for most countries in the world. Since all cosmetic ingredients in Europe have been banned from animal testing since March 2013, they may change in the future.
The safety of UV absorbers for sunscreens must be demonstrated in a wide range of toxicology research programs such as acute oral toxicity, chronic toxicity, dermal toxicity, embryo-fetal toxicity, light stimulation, transdermal absorption, photocarcinogenicity, medicine Generational dynamics and metabolism. UV absorbers for sunscreens are regulated worldwide as over-the-counter (OTC) drugs, quasi-drugs or cosmetics. All countries have positive list of UV absorbers, including the maximum concentration allowed for sunscreens. In most countries, including Europe and Japan, UV absorbers are prescribed as cosmetics; in the United States and Canada as over-the-counter drugs; in Australia as a therapeutic drug. Currently,a total of eight UV absorbers approved in all other regions are awaiting approval by the United States through the so-called Time and Range Application (TEA). By exhibiting at least five years of marketing experience in at least five countries outside the United States, the following eight UV absorbers have passed the first phase of the monograph, but they may not be foreseen by the US FDA for sunscreen (TEA submission year / qualification):
1)Amiloxate (2002)
2)Enzacamene (2002)
3)Octyl Triazolone(2002)
4)Bemotrizinol (2005)
5)Bisoctrizole (2005)
6)Iscotrizinol (2005)
7)Ecamsule (2007)
8)Drometrizole Trisiloxane (2009)
The PASS Alliance is monitoring the state of TEA and is supporting legislative initiatives to help the FDA solve the problem.
















